
When there is an imbalance between protons and neutrons, usually when the ratio of neutrons to protons is too low, the isotope will want to transform itself into a more stable form – a different atom. Isotopes that are larger and stable have slightly more neutrons than protons.Īn example of a stable isotope is carbon-12, which has six protons and six neutrons, for a total mass of 12g. When an isotope is small and stable, it contains close to an equal number of protons and neutrons.
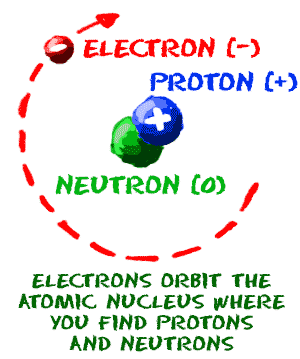
An isotope is stable when there is a balance between the number of neutrons and protons. They will not undergo radioactive decay and give off radiation. Both uranium-235 and uranium-238 are isotopes of uranium. Another example is uranium-235 which has 92 protons and 143 neutrons, and uranium 238 which has 92 protons and 146 neutrons. For example, there are three isotopes (or variants) of hydrogen: hydrogen-1 (one proton and no neutrons), hydrogen-2 or deuterium (one proton and one neutron), and hydrogen-3 which is called tritium (one proton and two neutrons). Nuclides of an element that have the same number of protons but not the same number of neutrons are called isotopes of that element. A molecule of water, for example, is formed of two atoms of hydrogen combined with one atom of oxygen (H 2O). When there is a significant imbalance between the number of neutrons and protons in a nucleus, the atom becomes unstable and in order to achieve stability, the atom may undergo a transformation or radioactive decay.Ītoms from one or more elements combine to form larger compounds, which are called molecules. For example, there are six protons in carbon therefore, its atomic number is 6 on the periodic table.Ītoms are stable when the number of neutrons and protons in the nucleus are balanced. The number of protons is unique to each element. It is the number of protons in the nucleus the atomic number that distinguishes each element. The negative electrons are attracted to the positive nucleus by an electrical force. The electrons, which carry a negative charge, move around the nucleus in clouds (or shells). The nucleus carries a positive charge protons are positively charged, and neutrons don't carry a charge. An atom consists of protons and neutrons, that make up the nucleus, and electrons that orbit the nucleus.
